Drug Overview
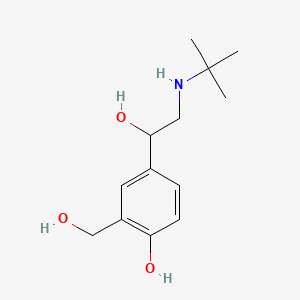
Chemical Name
α1-[[(1,1-dimethylethyl)amino]methyl]-4-hydroxy-1,3-benzenedimethanol hemisulfate
Generic Name
Albuterol
Drug Class
B-2 adrenoreceptor agonist
Description
Albuterol, also known as salbutamol, is approved by the U.S. Food and Drug Association (FDA) for treating and preventing acute or severe bronchospasm in patients with reversible obstructive airway disease, including exercise-induced bronchospasm. Albuterol is a commonly prescribed bronchodilator used to manage various respiratory conditions, including asthma and exercise-induced bronchospasm.
Mechanism of Action
The β2-adreneceptor agonist binds to the β2 receptor, which activates adenyl cyclase, resulting in the conversion of ATP to cyclic AMP (cAMP). This promotes bronchodilation and relieves symptoms experienced during an acute asthma episode.

Indications
- Asthma (mild to life-threatening)
- Reversible airway obstruction
- Exercise or allergen-induced bronchospasm
- Nocturnal asthma
- Premature labour (under specialist care)
Routes & Dosage
1. BY INHALATION (Aerosol / Powder / Nebuliser)
Acute Asthma | Exacerbation
- Child 1 month–4 yrs: 2.5 mg every 20–30 mins
- Child 5–11 yrs: 2.5–5 mg every 20–30 mins
- Child 12–17 yrs: 5 mg every 20–30 mins
- Adult: 5 mg every 20–30 mins (Oxygen-driven nebuliser preferred)
Moderate–Severe Asthma (Spacer Use)
- Child & Adult: 2–10 puffs every 10–20 mins (100 mcg/puff)
Bronchospasm Prophylaxis
- Adult: 200 mcg
- Child: 100–200 mcg up to 4×/day
2. BY MOUTH (Immediate-Release)
- Adult: 4 mg 3–4×/day (Max single dose: 8 mg)
- Use lower dose in elderly; inhalation preferred
3. BY INTRAVENOUS INJECTION (Severe cases only)
- Child 1–23 months: 5 mcg/kg over 5 min
- Child 2–17 yrs: 15 mcg/kg (Max 250 mcg) over 5 min
4. BY INTRAVENOUS INFUSION (Premature Labour)
- Adult: Start at 10 mcg/min; max 45 mcg/min
- Reduce rate after contractions stop; limit use to 48 hrs
| Formulation | Strengths Available | Adult Dosage | Pediatric Dosage |
|---|---|---|---|
| Inhaler (MDI/DPI) | 90 mcg/actuation (base)(=108 mcg albuterol sulfate) | 1–2 puffs every 4–6 hrs Max: 12 puffs/day Severe: 4–8 puffs every 20 min ×3, then q1–4 hrs |
Age ≥4 yrs: 1–2 puffs every 4–6 hrs Max: 12 puffs/day Exercise-induced: 2 puffs, 15–20 min before |
| Nebulizer Solution | 0.083%, 0.5%,0.63 mg/3 mL,1.25 mg/3 mL | 2.5 mg 2–3× daily or 1.25–5 mg every 4–8 hrs Severe: 2.5–5 mg every 20 min ×3, then q1–4 hrs |
<2 yrs: 0.2–0.6 mg/kg/day in divided doses ≥2 yrs: 2.5 mg every 6–8 hrs (Max: 10 mg/day) |
| Tablets (Immediate-Release) | 2 mg, 4 mg | 2–4 mg every 6–8 hrs Max: 32 mg/day |
≤6 yrs: 0.2–0.3 mg/kg/day ÷ q8 hrs (Max: 12 mg/day) 6–12 yrs: 2 mg q6–8 hrs (Max: 24 mg/day) >12 yrs: 2–4 mg q6–8 hrs (Max: 32 mg/day) |
| Tablets (Extended-Release) | 4 mg, 8 mg | 4–8 mg every 12 hrs Max: 32 mg/day |
6–12 yrs: 4 mg q12 hrs (Max: 24 mg/day) >12 yrs: 8 mg q12 hrs (Max: 32 mg/day) Contraindicated <6 yrs |
| Syrup | 2 mg / 5 mL | 2–4 mg every 6–8 hrs Max: 32 mg/day |
2–6 yrs: 0.1–0.2 mg/kg every 8 hrs 6–14 yrs: 2 mg q6–8 hrs >14 yrs: 2–4 mg q6–8 hrs (Max: 32 mg/day) |
Pharmacokinetic Profile
Absorption: Rapid after oral/inhaled use. Inhaled dose partly swallowed; lung uptake better with positive pressure breathing.
Tmax:
- Oral: 2.5–3 hrs
- Inhaled: ~3 hrs
Blood Levels:
- Oral (4–8 mg): ~23 ng/ml (unchanged), 50–100 ng/ml (total)
- Inhaled (80 μg): ~0.2 ng/ml (unchanged), ~1 ng/ml (total)
- IV (200 μg): ~1 ng/ml (unchanged), ~2 ng/ml (total)
Half-Life: 2–7 hours
Bioavailability: ~50% (oral); inhaled dose partly metabolized after swallowing.
Distribution: Vd ≈ 156 L
Metabolism: First-pass metabolism; exact pathways unknown.
Excretion:
- Oral: 75–95% urine, ~4% feces
- Inhaled: Up to 97% urine, ~11% feces; ~20% lost in air/device
- IV: ~80% urine, ~4% feces
- Metabolized: 50–60% (oral/inhaled), ~27% (IV)
Kinetics: First-order elimination
Steady State: Reached in 1–2 days with regular use
Side Effects
| Category | Frequency | Side Effects |
|---|---|---|
| General | Common/Very Common | Muscle cramps |
| Rare/Very Rare | Akathisia, Vasodilation | |
| Frequency Not Known | Metabolic changes, Myocardial ischaemia | |
| Specific (Inhalation) | Uncommon | Oral irritation, Throat irritation |
| Frequency Not Known | Lactic acidosis (high doses) | |
| Specific (Parenteral) | Uncommon | Pulmonary oedema |
| Frequency Not Known | Lactic acidosis (high doses), Vomiting |
Common Side Effects
- Tremor
- Headache
- Palpitations
- Muscle cramps
- Anxiety (More likely with oral/IV than inhaled route)
Tolerance develops with regular use
Serious Adverse Reactions (Rare but Possible)
- Myocardial ischemia (e.g., during labor infusion)
- Ventricular ectopics, angina (in patients with heart disease)
- Psychosis with excessive oral doses
Overdose
- Common symptoms: Tremor, flushing, palpitations, agitation
- Usually not fatal (even up to 240 mg)
- Management:
- Cardioselective β-blocker (avoid in asthma)
- Hypotension: Give fluids
- Labor cases: Add loop diuretic
- Monitor potassium (risk of hypokalemia)
Drug Interactions
| Interacting Drug/Class | Effect/Concern | Recommendation |
|---|---|---|
| TCAs / MAOIs | Enhanced cardiovascular effects when used with or within 2 weeks of stopping therapy | Use with caution, monitor cardiac status |
| Beta-blockers | May block bronchodilation effect of salbutamol; non-selective agents may cause bronchospasm | Avoid non-selective beta-blockers in asthma |
| Non–K⁺-sparing diuretics | ↑ Risk of hypokalemia, ECG changes, especially at high beta-agonist doses | Monitor electrolytes, use with caution |
| Digoxin | ↓ Serum digoxin levels (by 16–22%) when co-administered with albuterol | Monitor digoxin levels regularly |
Food Interactions
Caffeine-containing foods/beverages: May increase central nervous system stimulation and exacerbate tremors or palpitations.
Use in Pregnancy & Breastfeeding
Pregnancy
Salbutamol is categorized as Pregnancy Category C (U.S. FDA), which indicates that risk to the fetus cannot be ruled out. Although animal studies have shown adverse fetal effects, controlled studies in pregnant women are lacking. Therefore, it should only be used during pregnancy if the potential benefits outweigh the risks. It is sometimes used off-label to manage preterm labor due to its uterine relaxation effects.
Breast Feeding
Inhaled drugs for asthma can be taken as normal during breast-feeding.
Contraindications
Do not use in uncomplicated premature labour with conditions like abruptio placenta, antepartum haemorrhage, eclampsia, or cardiac disease history. Also contraindicated in placenta praevia, pulmonary hypertension, fetal death/infection, severe pre-eclampsia, or threatened miscarriage.
Unlicensed Use
- Oral (Syrup/Tablets): Not licensed for children under 2 years
- Intravenous (Injection/Infusion): Not licensed for children under 12 years. Use of undiluted injection via central venous catheter is also not licensed.
Toxicity Warning
- A dose of 1 mg/kg may be toxic in children under 6.
- Rarely, salbutamol can affect liver enzymes.
- Overdose is uncommon with inhalers but can still be dangerous.
Signs of Overdose
- Tremors (shaking)
- Fast or irregular heartbeat
- Low potassium levels (hypokalemia)
- High blood sugar (hyperglycemia)
- Lactic acidosis (can cause rapid breathing)
Emergency Management
- Stop using the medication immediately.
- Seek medical help for heart symptoms or severe weakness.
- Treatment includes:
- Supportive care
- Managing irregular heart rhythms
- Correcting potassium levels carefully
Always use salbutamol exactly as prescribed. Do not exceed the recommended dose.
Storage Conditions
| Form | Storage Conditions |
|---|---|
| Inhaler (MDI/DPI) | Store at below 30°C (86°F). Do not freeze. Keep away from direct sunlight and heat. Replace cap after use. |
| Nebulizer Solution | Store at room temperature (15–30°C). Protect from light. Do not use if solution becomes cloudy or discolored. |
| Tablets (IR/ER) | Store at 25°C (77°F); excursions permitted to 15–30°C. Keep tightly closed. |
| Syrup | Store at room temperature, preferably 15–30°C. Protect from moisture and light. |
| Injection/IV Solution | Store as per manufacturer's label (usually 2–8°C refrigerated). Do not freeze. Discard unused portions. |
Auxiliary Labels
Inhaler
- Shake well before use - Rinse mouth after use - Clean mouthpiece regularly - Carry at all times
Nebulizer
- For inhalation only - Use with nebulizer only - Discard after opening
Tablet/Syrup
- May cause dizziness or tremors - Take with food - Store in a cool, dry place
All Forms
- Use only as directed - Do not exceed dose - Keep out of reach of children
Directions for Administration
IV (Children):
- Infusion: Dilute to 200 mcg/mL with Glucose 5% or NaCl 0.9%.
- Injection: Dilute to 50 mcg/mL with Glucose 5%, NaCl 0.9%, or Water for Injections.
- Undiluted use via central line is unlicensed.
Inhalation (Nebulisation):
- Dilute with sterile NaCl 0.9% as per nebuliser type/time.
- Compatible with ipratropium bromide -- can be mixed.
IV (Adults):
- Bronchodilation: Dilute to 200 mcg/mL with Glucose 5% or NaCl 0.9%.
- Premature Labour:
- Syringe pump: 200 mcg/mL (Glucose 5%)
- Other methods: 20 mcg/mL
- Monitor fluid/electrolyte status closely.
Commonly Available Brands in Pakistan
Salbutamol is available in multiple formulations (inhalers, tablets, syrup, nebulizer solution) under various brand names in Pakistan:
Nebulizer: 5 mg/2.5 mL
Syrup: 2 mg/5 mL
Inhaler: 100 mcg/dose
Syrup: 2 mg/5 mL
References
- BNF 88 Sept 2024-March 2025
- The Pharmaceutical CODEX (11th Edition)
- Therapeutic Drugs Volume 1
- Pharma Guide
- NIH-Salbutamol
- Medscape-Salbutamol